Determination of Lincosamide Antibiotics Residues in Crab Tissues
-
摘要:
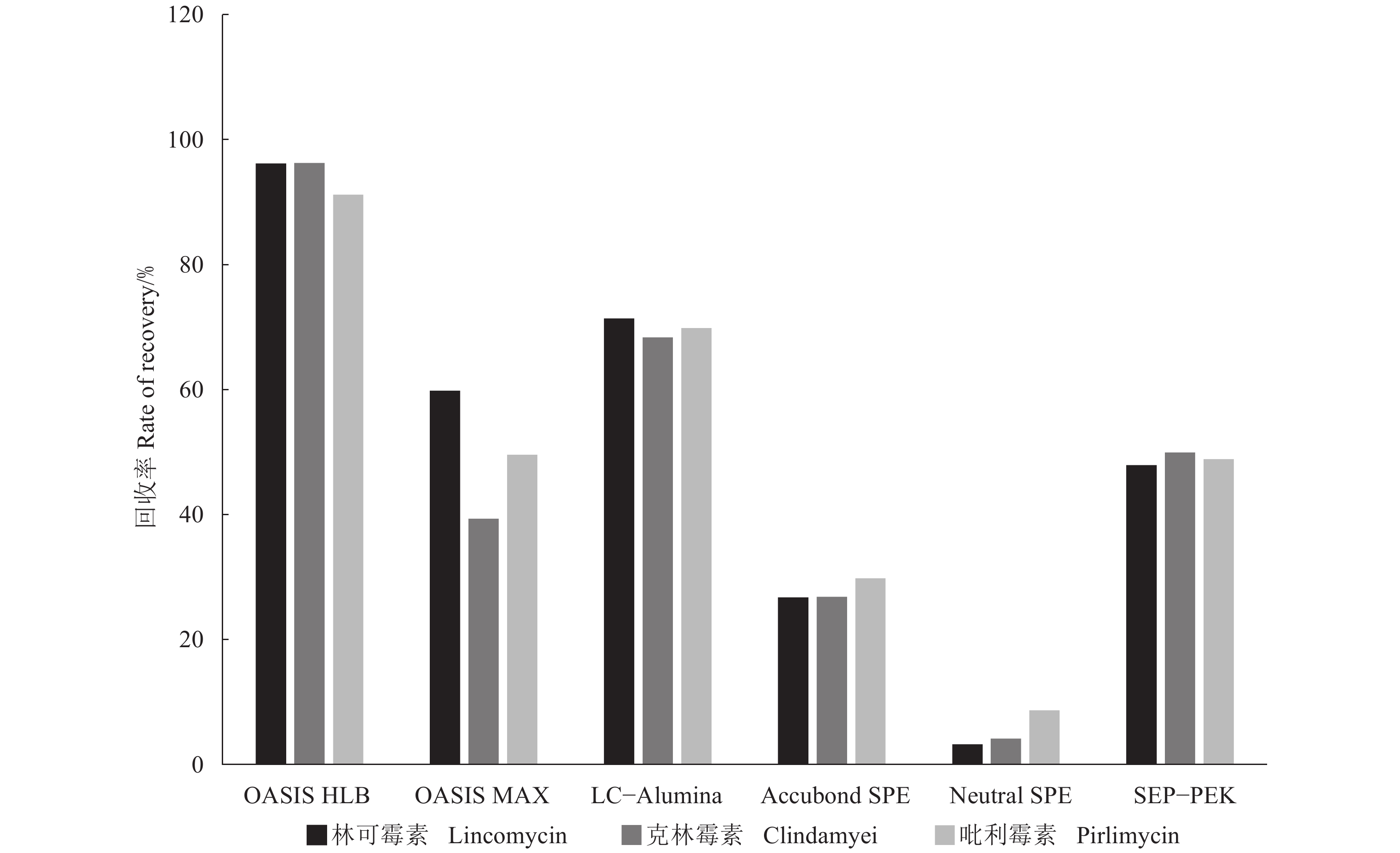
目的 基于固相萃取技术(SPE),建立一种可靠的高效液相色谱-串联质谱法测定河蟹样品中林可酰胺类抗生素药物的检测方法。 方法 以固相萃取柱为样品净化材料,考察其对河蟹肝胰腺样品的净化效果,优化色谱条件和质谱参数,并对方法进行评价。 结果 在河蟹肝胰腺基质中,OASIS® HLB SPE柱对3种药物的回收率最高。以InfinityLab poroshell 120 SB-C18(100 mm×2.7 mm,2.7 μm)柱为固定相,0.1%甲酸水(A相)和甲醇(B相)为流动相,采用多反应监测(MRM)模式对3种林可酰胺类药物进行检测和定量。3种目标药物为50 μg·L−1,方法线性良好,3种药物检出限为0.22~0.35 μg·kg−1,定量限为0.72~1.16 μg·kg−1,相关系数(R2)大于0.99。在添加量为5、10和50 μg·kg−1质量浓度的空白样品加标回收率试验中,3种林可酰胺类药物的绝对回收率为80.5%~99%,相对标准偏差为2.5%~5.2%。 结论 本LC-MS/MS方法提供了一种简便快捷的解决方案,以固相萃取技术为切入点,测定河蟹样品中3种林可酰胺类抗生素的残留,具有稳定可靠,重现性好的特点。 Abstract:Objective An HPLC-MS/MS method following an optimized solid phase extraction (SPE) procedure was established for the determination of lincosamide antibiotics in crab tissues. Method SPE column was used to separate and purify the substances from crab roe samples. Lincomycin, clindamycin, and pyrimycin were the target antibiotics to be detected by the methodology. Result Among the tested materials for the chemical separation, OASIS® HLB SPE column exhibited the highest recovery rate on the antibiotics. For the HPLC-MS/MS determination on the antibiotics, the gradient elution on an Infinity Lab poroshell 120 SB-C18 column (100 mm×2.7 mm, 2.7 μm) using 0.1% formic acid for the mobile phase A and methanol for the mobile phase B under the multi-reaction monitoring mode was applied. A measurement linearity within 50 μg·L−1 with a R2>0.99 was achieved. The assay performed with a recovery rate ranging 80.5%–99%, the relative standard deviation 2.5%–5.2%, the detection limit 0.22–0.35 μg·kg−1, and the quantitative limit 0.72–1.16 μg·kg−1. Conclusion The newly established methodology was stable and reliable in detecting the antibiotics in crab at a reduced cost. -
Key words:
- river crab /
- antibiotic residues /
- solid phase extraction /
- HPLC-MS/MS /
- lincosamide
-
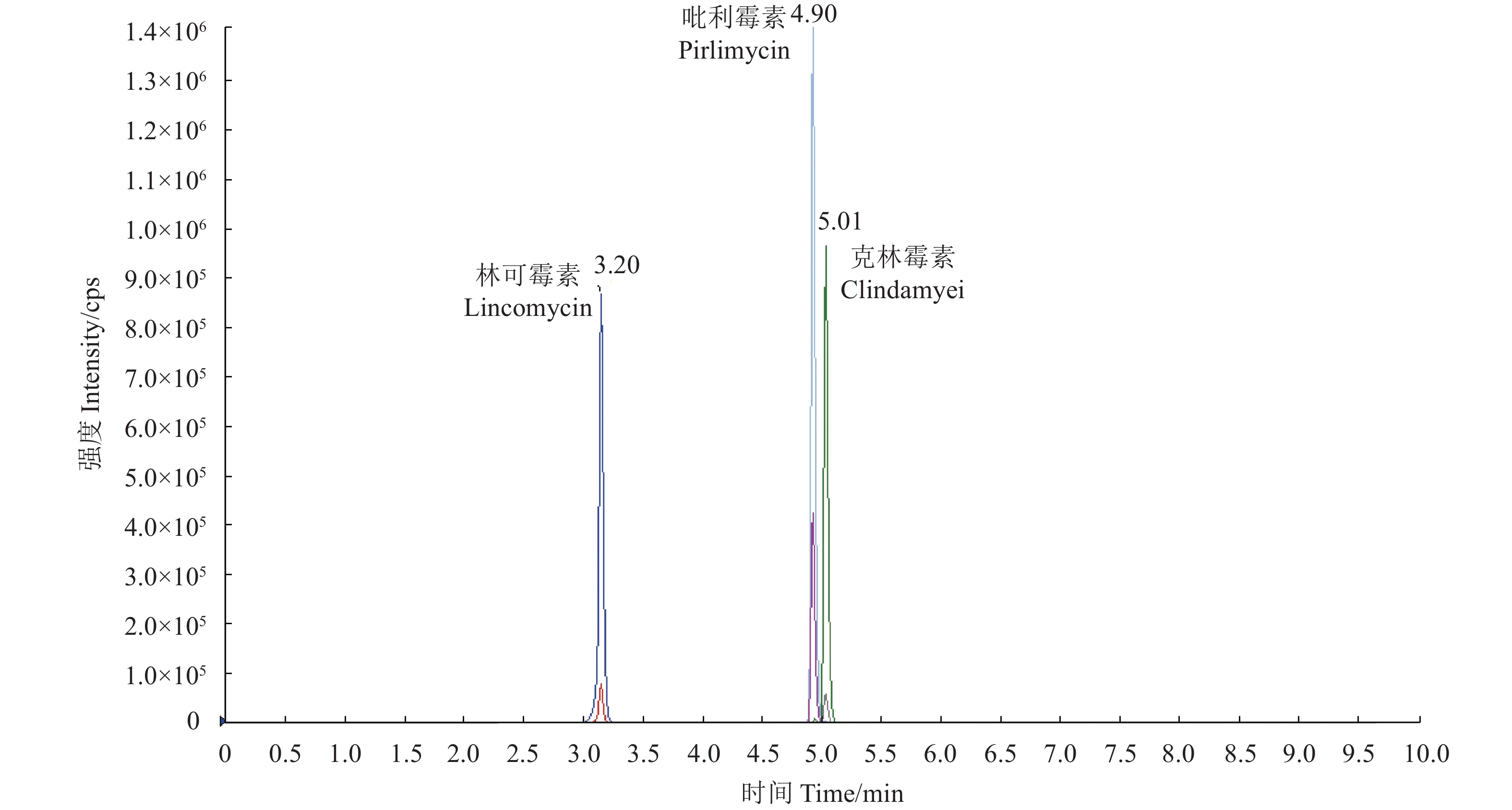
图 1 质量浓度为50 ng·L−1的 3 种林可酰胺类药物标准溶液的总离子流图
注:从左至右:峰 1,林可霉素;峰 2,吡利霉素;峰 3,克林霉素。 林可霉素、吡利霉素、克林霉素最高峰值分别为:8.7×105 cps、1.4×106 cps、9.6×105 cps。
Figure 1. Total ion chromatograms of 50 ng·L−1 standard solutions of 3 lincosamide antibiotics
Note: From left to right: peak 1, lincomycin; peak 2, Pirlimycin; peak 3, Clindamyei. The highest peaks of lincomycin,Pirlimycin and Clindamyei were 8.7×105 cps、1.4×106 cps、9.6×105 cps respectively.
表 1 流动相梯度洗脱程序
Table 1. Procedures of mobile phase gradient elution
时间
Time/minA相
Phase A/%B相
Phase B/%0.00 95 5 2.00 75 25 5.00 25 75 6.00 95 5 10.00 95 5 表 2 林可酰胺类药物测定的质谱参数
Table 2. Mass spectrometric parameters for lincosamide antibiotics determination
化合物名称
Compound
name定性离子对
Qualitative
Transition
(m/z)定量离子对
Quantitative
Transition
(m/z)碰撞能量
Collision
energy/eV去簇电压
Fragmentor/V林可霉素
Lincomycin407.3/126.1 407.3/126.1 34 85 407.3/359.4 26 85 吡利霉素
Pirlimycin411.3/112.1 411.3/112.1 32 85 411.3/363.3 25 85 克林霉素
Clindamyei425.3/126.2 425.3/126.2 34 85 425.3/377.2 27 85 表 3 方法的线性回归方程、相关系数、检出限和定量限
Table 3. Linear regression equation, correlation coefficient, detection limit, and quantitative limit of assay
目标物
Component回归方程
Regression equation线性范围
Linear range相关系数R2 检出限LOD/(μg·kg−1) 定量限LOQ/(μg·kg−1) 林可霉素 Lincomycin y=51216x+12618.2 0~50 0.99579 0.35 1.16 克林霉素 Clindamyei y=53764x+13338.2 0~50 0.99031 0.22 0.72 吡利霉素 Pirlimycin y=52491x+12978.3 0~50 0.99305 0.30 1.00 表 4 3种林可酰胺类药物的加标回收率及精密度(n=6)
Table 4. Recovery and precision of assay on 3 lincosamide antibiotics (n=6)
目标物
Component添加水平
Spiked levels/(μg·kg−1)回收率
Recovery/%相对标准偏差
RSD/%林可霉素 Lincomycin 5,10,50 99.0,95.4,96.7 2.8,3.2,2.5 克林霉素 Clindamyei 5,10,50 84.2,83.8,80.5 4.1,4.2,5.2 吡利霉素 Pirlimycin 5,10,50 91.5,89.7,88.5 3.5,3.7,3.9 -
[1] 孙玉杰, 杨淑慎, 张琪, 等. 丙氨酸对林可霉素发酵的影响 [J]. 山东化工, 2017, 46(16):9−10, 12. doi: 10.3969/j.issn.1008-021X.2017.16.005SUN Y J, YANG S S, ZHANG Q, et al. The effect of alanine on the fermentation of lincomycin [J]. Shandong Chemical Industry, 2017, 46(16): 9−10, 12.(in Chinese) doi: 10.3969/j.issn.1008-021X.2017.16.005 [2] ZHANG D Z, TANG Z J, LIU W. Biosynthesis of lincosamide antibiotics: Reactions associated with degradation and detoxification pathways play a constructive role [J]. Accounts of Chemical Research, 2018, 51(6): 1496−1506. doi: 10.1021/acs.accounts.8b00135 [3] 杨明, 伊鋆, 陈丹, 等. 氧化锌QuEChERS/高效液相色谱-串联质谱法测定蜂蜜中7种林可酰胺类与大环内酯类兽药残留 [J]. 分析测试学报, 2020, 39(8):974−979. doi: 10.3969/j.issn.1004-4957.2020.08.005YANG M, YI Y, CHEN D, et al. Determination of seven lincosamides and macrolides residues in honeys by high performance liquid chromatography-tandem mass spectrometry with ZnO QuEChERS [J]. Journal of Instrumental Analysis, 2020, 39(8): 974−979.(in Chinese) doi: 10.3969/j.issn.1004-4957.2020.08.005 [4] 何洁. 食品中林可酰胺类抗生素和黄曲霉毒素残留的酶联免疫分析方法研究[D]. 深圳: 深圳大学, 2018.HE J. Development of enzyme-linked immunosorbent assays for rapid detection of lincosamides and aflatoxins residues in foods[D]. Shenzhen, China: Shenzhen University, 2018. (in Chinese). [5] 胡楠, 裴科, 秦楠. HPLC法对林可霉素利多卡因凝胶中二组分及有关物质的含量测定 [J]. 北方药学, 2020, 17(3):1−2. doi: 10.3969/j.issn.1672-8351.2020.03.001HU N, PEI K, QIN N. Determination of two components and related substances in lincomycin lidocaine gel by HPLC [J]. Journal of North Pharmacy, 2020, 17(3): 1−2.(in Chinese) doi: 10.3969/j.issn.1672-8351.2020.03.001 [6] 何方洋, 冯才伟, 杜美红, 等. 一种林可霉素的磁免疫化学发光检测试剂盒及其应用: CN104897896B[P]. 2016-08-24. [7] 王可, 曹倩玉, 赵灵芝, 等. 高效液相色谱串联质谱法测定禽蛋中异丙嗪和氯丙嗪 [J]. 食品工业, 2020(3):299−302.WANG K, CAO Q Y, ZHAO L Z, et al. Determination of promethazine and chlorpromazine in poultry eggs by high performance liquid chromatography tandem mass spectrometry [J]. The Food Industry, 2020(3): 299−302.(in Chinese) [8] 王静静. 复杂基质食品样品中多种农药残留和兽药残留检测方法的研究[D].新疆: 新疆大学, 2017.WANG J J. Determination of multiple pesticides and veterinary drugs residues in complicated foodstuff samples[D].Xinjiang: Xinjiang University, 2017. [9] 侯卓. 头孢类抗生素的固相萃取-高效毛细管电泳方法建立与研究[D]. 上海: 上海交通大学, 2018.HOU Z. Establishment and research of solid-phase extraction-capillary electrophoresis method for cephalosporins[D]. Shanghai: Shanghai Jiaotong University, 2018. (in Chinese). [10] 洪博, 陈刚, 赵超阳, 等. 固相萃取结合高效液相色谱法测定甘草中6种有效成分的含量 [J]. 中国医药导报, 2020, 17(36):126−129.HONG B, CHEN G, ZHAO C Y, et al. Determination on the contents of 6 active ingredients in Glycyrrhizae Radix et Rhizoma by solid phase extraction and high performance liquid chromatography [J]. China Medical Herald, 2020, 17(36): 126−129.(in Chinese) [11] BOSACCHI, M, TODOSOV, et al. Synthetic lincosamide detoxification genes for highly efficient plant transformation [J]. Vitro Cell Dev An, 2018. [12] 江竹莲, 张伟, 刘绿叶. 柱切换二维色谱除盐质谱联用鉴定阿莫西林中杂质 [J]. 山东化工, 2015, 44(2):59−62, 66. doi: 10.3969/j.issn.1008-021X.2015.02.019JIANG Z L, ZHANG W, LIU L Y. Determination of impurities in amoxicillin by desalt mobile phase with 2D column switch and LC-MS [J]. Shandong Chemical Industry, 2015, 44(2): 59−62, 66.(in Chinese) doi: 10.3969/j.issn.1008-021X.2015.02.019 [13] 邹汉法, 陈津, 王方军. 液相色谱-质谱联用对分析物进行高效分离鉴定的方法: CN105987964B[P]. 2018-05-15. [14] 马晓斐, 宋炜, 梁天佐, 等. 微波辅助萃取-超高效液相色谱-质谱法测定食品用纸容器中双酚A [J]. 食品科学, 2014, 35(16):165−169. doi: 10.7506/spkx1002-6630-201416032MA X F, SONG W, LIANG T Z, et al. Determination of bisphenol A in paper food containers by ultra performance liquid chromatography and tandem mass spectrometry [J]. Food Science, 2014, 35(16): 165−169.(in Chinese) doi: 10.7506/spkx1002-6630-201416032 [15] KANAI M, IIDA A, NAGAOKA Y, et al. Fungal metabolites. XXI. 1 Characteristics of low energy collision induced dissociation of [M + 2H]2+, [M + H + Na]2+ and [M + 2Na]2+ of peptaibols using electrospray ionization mass spectrometry [J]. Journal of Mass Spectrometry, 1996, 31(2): 177−183. doi: 10.1002/(SICI)1096-9888(199602)31:2<177::AID-JMS286>3.0.CO;2-M [16] 林涛, 李泳波, 陈兴连, 等. 毛叶枣中多菌灵、吡虫啉和啶虫脒消解动态研究 [J]. 热带作物学报, 2018, 39(8):1630−1635. doi: 10.3969/j.issn.1000-2561.2018.08.024LIN T, LI Y B, CHEN X L, et al. Degradation dynamics of carbendazim, imidacloprid and acetamiprid in Ziziphus mauritiana [J]. Chinese Journal of Tropical Crops, 2018, 39(8): 1630−1635.(in Chinese) doi: 10.3969/j.issn.1000-2561.2018.08.024 [17] KONERMANN L. Addressing a common misconception: Ammonium acetate as neutral pH “buffer” for native electrospray mass spectrometry [J]. Journal of the American Society for Mass Spectrometry, 2017, 28(9): 1827−1835. doi: 10.1007/s13361-017-1739-3 [18] BUSSY U, JURVA U, BOISSEAU R, et al. Unexpected benzimidazole ring formation from a quinoneimide species in the presence of ammonium acetate as supporting electrolyte used in the coupling of electrochemistry with mass spectrometry [J]. Rapid Communications in Mass Spectrometry, 2015, 29(5): 456−460. doi: 10.1002/rcm.7122 [19] GUO Y R, GU S Q, WANG X C, et al. Nutrients and non-volatile taste compounds in Chinese mitten crab by-products [J]. Fisheries Science, 2015, 81(1): 193−203. doi: 10.1007/s12562-014-0816-9 [20] 刘志强, 冯建彬, 邱高峰. 中华绒螯蟹卵细胞透明液的配方 [J]. 水产学报, 2019, 43(4):852−857.LIU Z Q, FENG J B, QIU G F. Clearing solution's ingredients for oocyte in Chinese mitten crab(Eriocheir sinensis) [J]. Journal of Fisheries of China, 2019, 43(4): 852−857.(in Chinese) [21] AYISI C L, APRAKU A, AFRIYIE G. A review of probiotics, prebiotics, and synbiotics in crab: Present research, problems, and future perspective [J]. Journal of Shellfish Research, 2017, 36(3): 799−806. doi: 10.2983/035.036.0329 [22] STEINER D, KRSKA R, MALACHOVÁ A, et al. Evaluation of matrix effects and extraction efficiencies of LC-MS/MS methods as the essential part for proper validation of multiclass contaminants in complex feed [J]. Journal of Agricultural and Food Chemistry, 2020, 68(12): 3868−3880. doi: 10.1021/acs.jafc.9b07706 [23] FANG N B, YU S G, RONIS M J, et al. Matrix effects break the LC behavior rule for analytes in LC-MS/MS analysis of biological samples [J]. Experimental Biology and Medicine (Maywood, N J), 2015, 240(4): 488−497. doi: 10.1177/1535370214554545 [24] KACZYŃSKI P. Clean-up and matrix effect in LC-MS/MS analysis of food of plant origin for high polar herbicides [J]. Food Chemistry, 2017, 230: 524−531. doi: 10.1016/j.foodchem.2017.03.091 [25] 张协光, 肖伟敏, 朱丽, 等. 超高效液相色谱-线性离子阱-高分辨质谱同步检测果蔬及饮料中花青素 [J]. 分析试验室, 2019, 38(10):1199−1204.ZHANG X G, XIAO W M, ZHU L, et al. Simultaneous analysis of anthocyanins in fruit, vegetable and beverage by ultra-high performance liquid chromatography coupled to a linear ion trap-orbitrap high resolution mass spectrometer [J]. Chinese Journal of Analysis Laboratory, 2019, 38(10): 1199−1204.(in Chinese) [26] TRUBACA-BOGINSKA A, ACTINS A, ŠVINKA R, et al. The matrix effect and application of the multi-parameter optimization method for X-ray spectrometric determination of the quantitative composition of clays [J]. Key Engineering Materials, 2018, 788: 108−113. doi: 10.4028/www.scientific.net/KEM.788.108 [27] YUAN J H, ZHAN X C, HU M Y, et al. Characterization of matrix effects in microanalysis of sulfide minerals by laser ablation-inductively coupled plasma-mass spectrometry based on an element pair method [J]. Guang Pu Xue Yu Guang Pu Fen Xi, 2015, 35(2): 512−518. [28] 吴陈涛, 金敏敏, 邓泽融, 等. 高效液相色谱-串联质谱法测定淡水养殖池塘水体及鱼虾体内的藻毒素 [J]. 分析科学学报, 2017, 33(4):557−561.WU C T, JIN M M, DENG Z R, et al. Determination of microcystins in fresh aquaculture water and aquatic products by high performance liquid chromatography-tandem mass spectrometry [J]. Journal of Analytical Science, 2017, 33(4): 557−561.(in Chinese) [29] ZHANG S, GUO Y M, YAN Z Y, et al. A selective biomarker for confirming nitrofurazone residues in crab and shrimp using ultra-performance liquid chromatography-tandem mass spectrometry [J]. Analytical and Bioanalytical Chemistry, 2015, 407(30): 8971−8977. doi: 10.1007/s00216-015-9058-7 -






 下载:
下载: