Effect of TOR Inhibitor Rapamycin on Growth, Development, and Autophagy of Peronophythora litchii
-
摘要:
目的 明确TOR(Target of Rapamycin)信号通路抑制剂雷帕霉素对荔枝霜疫霉生长发育、致病性及自噬的影响。 方法 采用不同浓度的TOR抑制剂雷帕霉素处理荔枝霜疫霉菌,观察不同浓度雷帕霉素对荔枝霜疫霉的菌丝生长及形态、孢子囊产生数量、游动孢子释放和致病性的影响,并通过丹酰戊二胺(MDC)染色观察细胞自噬。 结果 雷帕霉素处理后,荔枝霜疫霉的菌丝生长明显受到抑制,半最大效应浓度(EC50)值为29.18 ng·mL−1,随着雷帕霉素浓度的增加,菌丝生长抑制不同程度增加;当雷帕霉素深度为25.0 ng·mL−1时,菌落生长抑菌率为45.3%,孢子囊产生数量为8.0×104 个·mL−1,仅为对照组的27.9%;雷帕霉素处理能促进游动孢子的释放;同时严重影响荔枝霜疫霉菌的致病性。通过自噬体观察表明,雷帕霉素处理后荔枝霜疫霉菌丝中自噬体的数量显著增加,促进荔枝霜疫霉的细胞自噬。 结论 TOR抑制剂雷帕霉素通过调控荔枝霜疫霉的细胞自噬,从而影响病菌的生长发育及致病性,研究结果为荔枝霜疫霉的致病机制提供科学依据。 Abstract:Objective Effects of the target of rapamycin (TOR) inhibitor rapamycin on the growth, development, pathogenicity, and autophagy of Peronophythora litchi were studied. Method The mycelial growth, sporangial production, zoospore release, and pathogenicity of the P. litchi after being treated by the TOR inhibitor rapanmycin in varying concentrations were determined, while the mycelial autophagy observed under a microscope with MDC staining. Result Rapamycin exhibited significant inhibitory effect on the mycelial growth of P. litchii with an EC50 value of 29.18 ng·mL−1 and varying extents of increases with higher dosages. When treated with 25.0 ng·mL−1 rapamycin, the inhibition rate was 45.3% and the sporulation at 8.0×104 sporangia·mL−1 (which was 27.9% of control), while the zoospore release increased, but the virulence of P. litchii significantly reduced. The MDC-stained autophagosomes significantly increased as well. Conclusion The TOR signaling pathway inhibitor rapamycin played an important role in the mycelial growth, development, and virulence of P. litchii. -
Key words:
- Peronophythora litchii /
- rapamycin /
- TOR inhibitor /
- growth and development /
- autophagy
-
图 1 雷帕霉素对荔枝霜疫霉生长的影响
①A:不同浓度雷帕霉素处理后荔枝霜疫霉的生长情况;B:不同含量雷帕霉素处理后荔枝霜疫霉影响的菌落直径。②a:CK,b:DMSO,c~f分别表示雷帕霉素含量为12.5、25、50、100 ng·mL−1。③图中数据为平均数±标准差,**表示经单因素方差分析法检验在P<0.01水平差异显著,图2~4同。
Figure 1. Effect of rapamycin on growth of P. Litchii
①A: Growth of P. litchii as affected by rapamycin dosages; B: Colony diameter of P. litchii treated by varied rapamycin dosages. ②a:CK,b:DMSO,c~f respectively represent rapamycin content 12.5、25、50、100 ng·mL−1。③Data are mean±SE; ** indicates significant difference at P<0.01 by one-way ANOVA; same for Figs. 2-4.
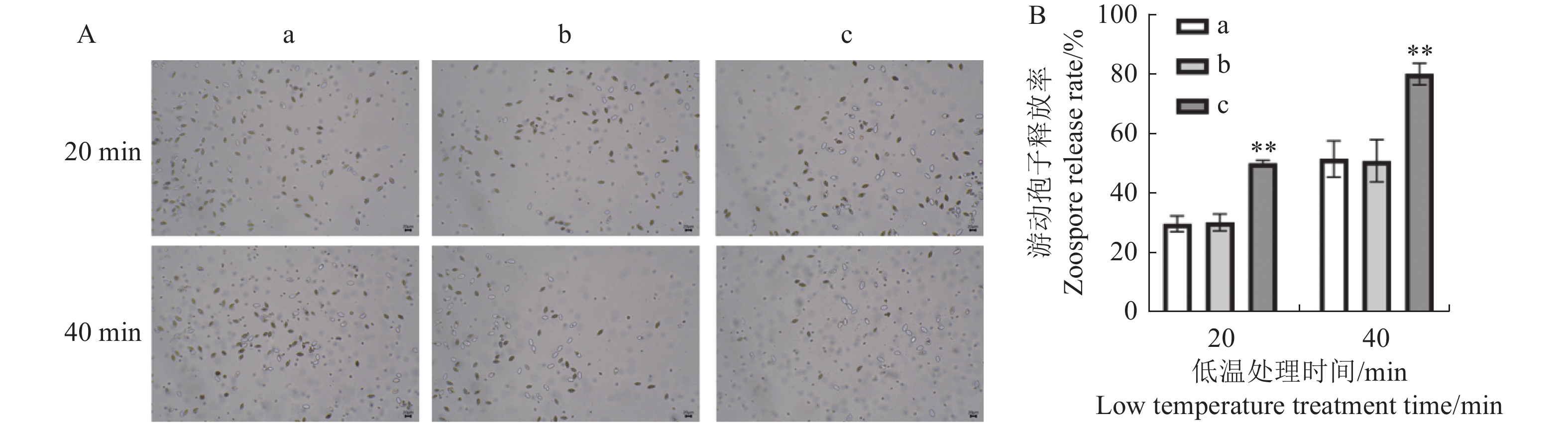
图 2 雷帕霉素对荔枝霜疫霉产孢的影响
①A:雷帕霉素处理后荔枝霜疫霉孢子囊数量的显微观察;B:雷帕霉素处理后荔枝霜疫霉孢子囊数量。②a:CK,b:DMSO,c:25 ng·mL−1雷帕霉素处理,图3同。
Figure 2. Effect of rapamycin on production of P. litchii sporangia
①A: Rapamycin-treated P. litchii sporangia under microscope; B: sporangia count.②a:CK,b:DMSO,c:25 ng·mL−1 Rapamycin treatment, same for Figs. 3.
图 4 雷帕霉素对荔枝霜疫霉致病性的影响
①A:雷帕霉素处理的荔枝霜疫霉叶片致病性;B:荔枝叶片处理病斑面积比例;C:雷帕霉素处理的荔枝霜疫霉果实致病性;D:荔枝果实处理病斑面积比例。②a:CK,b:DMSO,c:25 ng·mL−1,MOCK:空白对照。
Figure 4. Effect of rapamycin on pathogenicity of P. litchii
①A: Pathogenicity of rapamycin-treated P. litchii on lychee leaf; B: proportion of lesion area on lychee leaf; C: pathogenicity of rapamycin-treated P. litchii on lychee fruit; D: proportion of lesion area on lychee fruit.②a:CK,b:DMSO,c:25 ng·mL−1,MOCK:Blank control。
-
[1] 蔡学清, 林娜, 陈炜, 等. 荔枝霜疫霉的生物学特性 [J]. 热带作物学报, 2009, 30(9):1226−1231. doi: 10.3969/j.issn.1000-2561.2009.09.002CAI X Q, LIN N, CHEN W, et al. Biological characterisitics of Peronophthora litchii [J]. Chinese Journal of Tropical Crops, 2009, 30(9): 1226−1231.(in Chinese) doi: 10.3969/j.issn.1000-2561.2009.09.002 [2] 孔广辉, 冯迪南, 李雯, 等. 荔枝霜疫病的研究进展 [J]. 果树学报, 2021, 38(4):603−612.KONG G H, FENG D N, LI W, et al. Research progress in studies on the downy blight disease in litchi [J]. Journal of Fruit Science, 2021, 38(4): 603−612.(in Chinese) [3] 杨德东. 荔枝霜疫霉病的发生与防控 [J]. 中国热带农业, 2015, 65(4):58−59. doi: 10.3969/j.issn.1673-0658.2015.04.016YANG D D. Occurrence and control of Peronophythora lichii [J]. China Tropical Agriculture, 2015, 65(4): 58−59.(in Chinese) doi: 10.3969/j.issn.1673-0658.2015.04.016 [4] WANG H, SUN H, STAMMLER G, et al. Generation and characterization of isolates of Peronophythora litchii resistant to carboxylic acid amide fungicides [J]. Phytopathology, 2010, 100(5): 522−527. doi: 10.1094/PHYTO-100-5-0522 [5] WEN X, KLIONSKY D J. An overview of macroautophagy in yeast [J]. Journal of Molecular Biology, 2016, 428: 1681−1699. doi: 10.1016/j.jmb.2016.02.021 [6] REGGIORI F, KLIONSKY D J. Autophagic processes in yeast: Mechanism, machinery and regulation [J]. Genetics, 2013, 194(2): 341−361. doi: 10.1534/genetics.112.149013 [7] WULLSCHLEGER S, LOEWITH R, HALL M N. TOR signaling in growth and metabolism [J]. Cell, 2006, 124(3): 471−484. doi: 10.1016/j.cell.2006.01.016 [8] NODA T. Regulation of autophagy through TORC1 and mTORC1 [J]. Biomolecules, 2017, 7(3): E52. [9] KIM Y C, GUAN K L. mTOR: A pharmacologic target for autophagy regulation [J]. The Journal of Clinical Investigation, 2015, 125(1): 25−32. doi: 10.1172/JCI73939 [10] ZHU X M, LI L, WU M, et al. Current opinions on autophagy in pathogenicity of fungi [J]. Virulence, 2019, 10(1): 481−489. doi: 10.1080/21505594.2018.1551011 [11] KASAHARA K. Physiological function of FKBP12, a primary target of rapamycin/FK506: a newly identified role in transcription of ribosomal protein genes in yeast [J]. Current Genetics, 2021, 67(3): 383−388. doi: 10.1007/s00294-020-01142-3 [12] LI L, ZHU T, SONG Y, et al. Target of rapamycin controls hyphal growth and pathogenicity through FoTIP4 in Fusarium oxysporum [J]. Molecular Plant Pathology, 2021, 22(10): 1239−1255. doi: 10.1111/mpp.13108 [13] BALDAUF S L, ROGER A J, WENK-SIEFERT I, et al. A Kingdom-level phylogeny of eukaryotes based on combined protein data [J]. Science, 2000, 290(5493): 972−977. doi: 10.1126/science.290.5493.972 [14] JIANG L, SITU J, DENG Y Z, et al. PlMAPK10, a mitogen-activated protein kinase (MAPK) in Peronophythora litchii, is required for mycelial growth, sporulation, laccase activity, and plant infection [J]. Frontiers in Microbiology, 2018, 9: 426. doi: 10.3389/fmicb.2018.00426 [15] STEWART E L, HAGERTY C H, MIKABERIDZE A, et al. An improved method for measuring quantitative resistance to the wheat pathogen Zymoseptoria tritici using high-throughput automated image analysis [J]. Phytopathology, 2016, 106(7): 782−788. doi: 10.1094/PHYTO-01-16-0018-R [16] LUO Q, WANG F X, ZHONG N Q, et al. The role of autophagy during development of the oomycete pathogen Phytophthora infestans [J]. Journal of Genetics and Genomics, 2014, 41(4): 225−228. doi: 10.1016/j.jgg.2014.03.004 [17] SAXTON R A, SABATINI D M. mTOR Signaling in growth, metabolism, and disease [J]. Cell, 2017, 168(6): 960−976. doi: 10.1016/j.cell.2017.02.004 [18] 朱书生, 卢晓红, 陈磊, 等. 羧酸酰胺类(CAAs)杀菌剂研究进展 [J]. 农药学学报, 2010, 12(1):1−12. doi: 10.3969/j.issn.1008-7303.2010.01.01ZHU S S, LU X H, CHEN L, et al. Research advances in carboxylic acid amide fungicides [J]. Chinese Journal of Pesticide Science, 2010, 12(1): 1−12.(in Chinese) doi: 10.3969/j.issn.1008-7303.2010.01.01 [19] IADEVAIA V, LIU R, PROUD C G. mTORC1 signaling controls multiple steps in ribosome biogenesis [J]. Seminars in Cell & Developmental Biology, 2014, 36: 113−120. [20] CORNU M, ALBERT V, HALL M N. mTOR in aging, metabolism, and cancer [J]. Current Opinion in Genetics & Development, 2013, 23(1): 53−62. [21] XIONG F, LIU M, ZHUO F, et al. Host-induced gene silencing of BcTOR in Botrytis cinerea enhances plant resistance to grey mould [J]. Molecular Plant Pathology, 2019, 20(12): 1722−1739. doi: 10.1111/mpp.12873 [22] YU F, GU Q, YUN Y, et al. The TOR signaling pathway regulates vegetative development and virulence in Fusarium graminearum [J]. New Phytologist, 2014, 203(1): 219−232. doi: 10.1111/nph.12776 [23] 魏宁, 赵玉兰, 田菲菲, 等. TOR途径对玉米大斑病菌发育调控作用的研究 [J]. 植物病理学报, 2019, 49(3):391−398.WEI N, ZHAO Y L, TIAN F F, et al. Regulatory effect of TOR signal pathway on the development of Setosphaeria turcica [J]. Acta Phytopathologica Sinica, 2019, 49(3): 391−398.(in Chinese) [24] ZHANG S, KHALID A R, GUO D, et al. TOR inhibitors synergistically suppress the growth and development of Phytophthora infestans, a highly destructive pathogenic oomycete [J]. Frontiers in Microbiology, 2021, 12: 596874. doi: 10.3389/fmicb.2021.596874 -







 下载:
下载: