Isolation, identification and pathogenicity of Mycoplasma synoviae strains from Fujian to 7-day-old SPF chickens
-
摘要:
目的 了解福建地区滑液囊支原体(Mycoplasma synoviae,MS)临床感染菌株的进化关系以及致病能力。 方法 对福建地区疑似MS感染鸡群的上颚裂和气管拭子样品进行病原检测、分离和鉴定,获得MS临床分离株,并进行vlhA基因进化树分析;选取其中6株MS分离株,通过点眼滴鼻途径感染7日龄SPF鸡,观察感染后的临床症状和解剖病理变化、气管组织病理损伤、气管病原再分离以及MS抗体阳性率,比较不同菌株的致病力和水平传播能力。 结果 气管拭子的MS检出率显著高于上颚裂拭子。通过分离、鉴定共获得9株MS分离株,遗传进化关系分析表明vlhA基因存在着多样性,不同分离株具有不同的进化来源。根据vlhA基因的进化关系,选择6株MS分离株感染7日龄SPF鸡,未观察到明显的临床症状;临床解剖发现,感染HI株14 d、感染 HI和SD6株21 d各有1羽鸡出现了气囊炎,而其余各鸡以及同居对照鸡均未发现有明显病理变化。气管组织病理学分析发现,不同MS分离株在感染后7、14、21 d的病理损伤能力存在显著差异;气管MS病原再分离结果显示,不同分离株在气管的定殖和复制能力存在明显差异,并且SD19和SD6株还具有较强的水平传播能力。MS抗体检测发现仅在感染后21 d SD19株感染组出现1羽感染鸡和1羽同居对照鸡抗体转阳。 结论 气管样品更适合MS的检测和分离,福建MS分离株不易引起7日龄SPF鸡临床解剖病理变化,但可在气管中持续存在和定殖,造成病理损伤,且不同菌株之间存在显著差异。本研究通过对不同MS菌株在7日龄SPF鸡中的致病力和水平传播能力的比较为后续开展滑液囊支原体病的防控技术研究奠定了基础。 Abstract:Objective To understand the evolutionary relationship and pathogenicity of Mycoplasma synoviae (MS) clinical infection strains in Fujian Province. Method MS detection and isolation were performed from cleft palate and tracheal swab samples of chickens suspected of MS infection to obtain MS clinical isolates, and the vlhA gene phylogenetics of MS isolates were carried out. And also, the pathogenicity and horizontal transmission ability of MS isolates from Fujian province were evaluated using 7-day-old SPF chickens infected by eye and nasal route, including clinical symptoms and anatomical pathology, histopathology of trachea, MS re-isolation of trachea and MS antibody. Result The positive rate of MS in tracheal swabs was significantly higher than that in cleft palate swabs. A total of 9 MS isolates showed that there was diversity in the vlhA gene, and different isolates had different evolutionary sources. According to the phylogenetics of vlhA gene, 6 MS isolates were selected to infect 7-day-old SPF chickens, no obvious clinical symptoms were caused in SPF chickens from 1 to 21 days after infection. It was found that one chicken had mild air sacculitis in HI strain group at 14 and 21 days after infection and in SD6 strain group at 21 days after infection via necropsy, and other chickens including cohabitation chickens did not have obvious clinical anatomical and pathological changes. Histopathological analysis showed that there were significant differences in the tracheal lesion to different MS isolates after 7, 14, and 21 days of infection. The results showed that re-isolations of MS from trachea were with significant differences to different MS isolates, and SD19 and SD6 strains had strong horizontal transmission ability. MS antibody detection found that only 1 infected chicken and 1 cohabiting control chicken in the SD19 infection group turned positive for antibodies at 21 days after infection. Conclusion Tracheal samples were more suitable for the detection and isolation of MS. MS isolates were rather difficult causing clinicopathological changes in 7-day old SPF chickens, and also MS isolates could present and colonize in the trachea to cause histopathological lesion, and there were significant differences within different isolates. Therefore, the results laid the foundation for subsequent research on the prevention and control of Mycoplasma synoviae infection. -
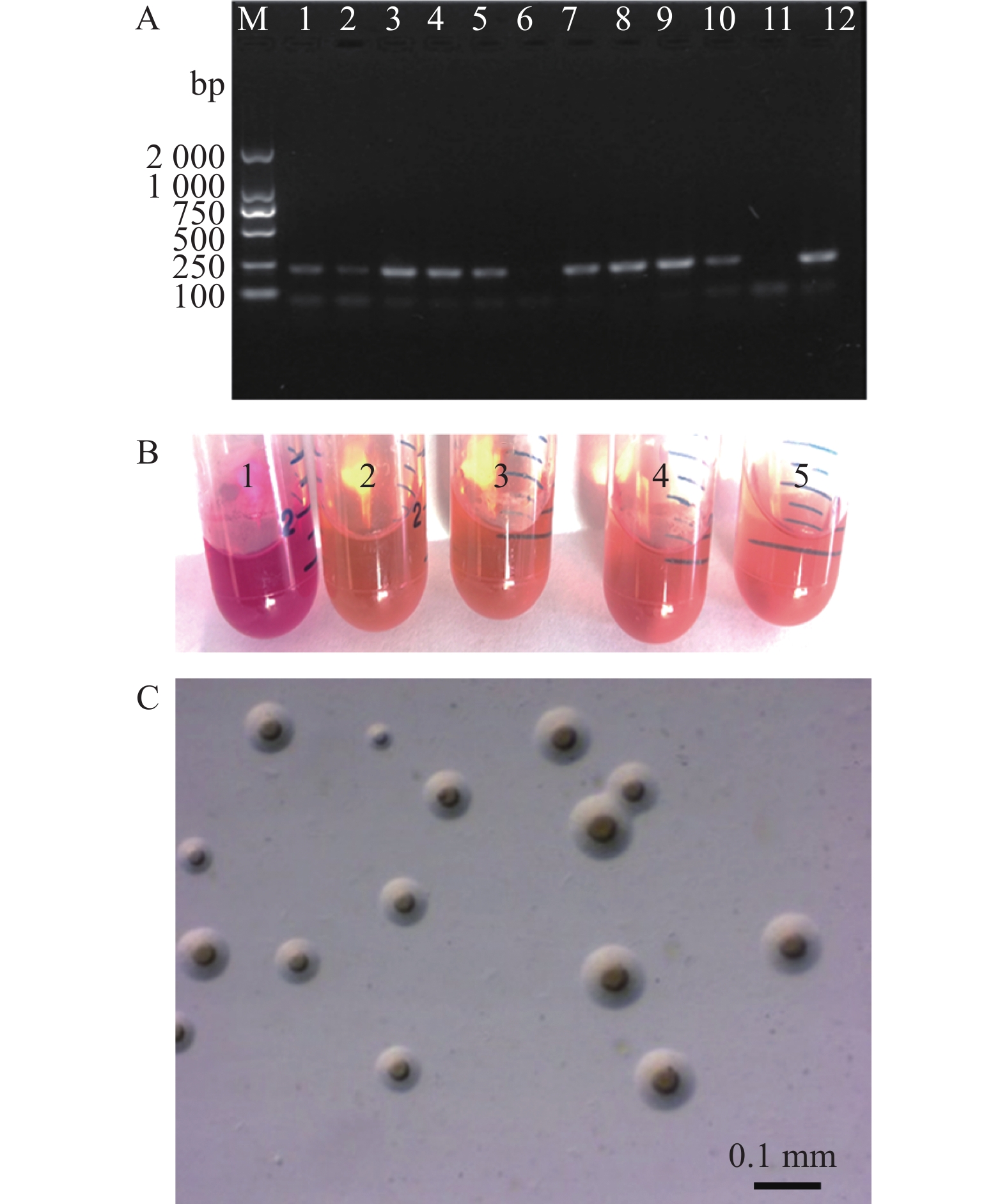
图 1 MS分离株的分离与鉴定
A——KM2液体培养基中培养18~20 h的原代培养物的PCR检测结果,M:DNA标记物,1 ~ 10:临床样品扩增结果,11:PCR阴性对照,12:PCR阳性对照。B——MS分离菌株在KM2液体培养基中的生长情况,1:玫瑰红颜色的培养基(阴性对照),2 ~ 5:培养3 d后的培养基呈现橘黄色。C——滑液囊支原体HI株在KM2琼脂上呈“肚脐状”或“煎蛋状”菌落形态(50×)。
Figure 1. Isolation and identification of MS isolates
A: Specificity of the PCR assay using specific primers performed on the primary culture in a KM2 medium for 18 to 20 h; M: DNA marker; 1 to10: cultures of clinical samples; 11: negative control; 12: positive control. B: Growth of MS isolates in KM2 medium; 1: Rose red color of the medium as negative control, 2 to 5: Orange color of medium cultivated MS strain after 3 days, C: Colonies of Mycoplasma synoviae HI strain on the KM2 agar (50×).
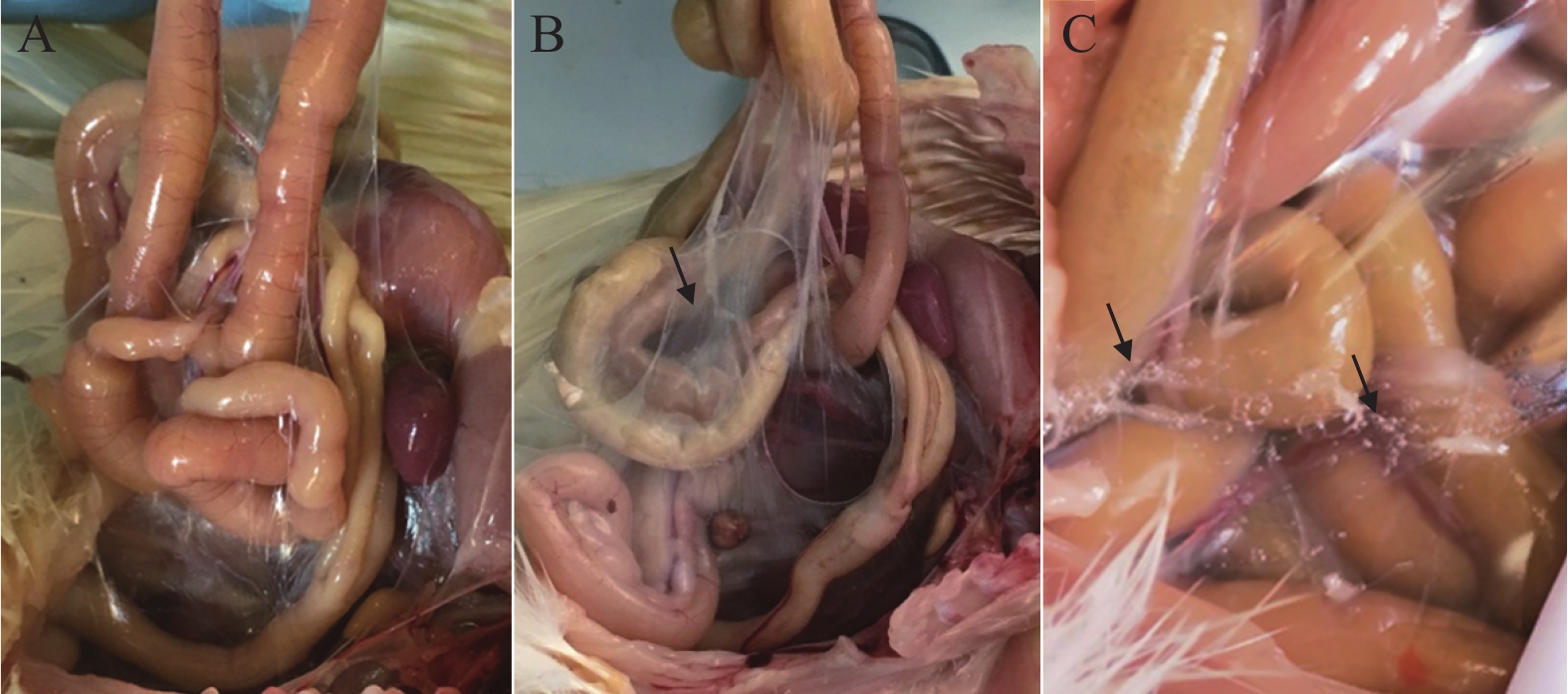
图 3 MS分离株HI感染SPF鸡14 d(B)和21 d(C)后气囊临床病理变化
A:正常气囊薄而透明;B:气囊增厚,透明度下降(黑色箭头);C:气囊表面有大量气泡(黑色箭头)。
Figure 3. Clinical anatomical and pathological changes of SPF chickens air sac infected with HI strain for 14 (B) and 21 (C) days
A: Normal air sac is thin and transparent; B: air sac is thickened (black arrow); C: air sac has foam on the surface (black arrow).
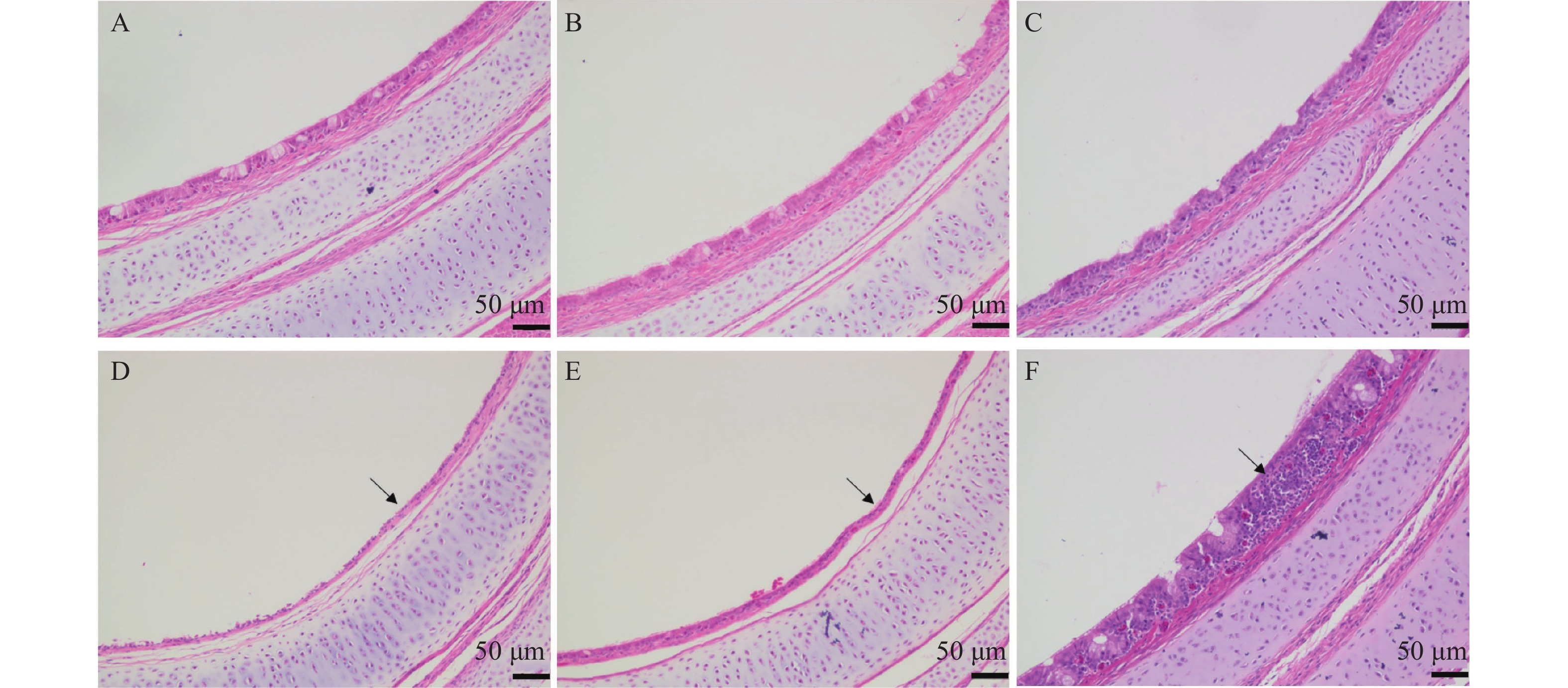
图 4 MS HI株感染SPF鸡7、14和21 d后气管组织病理学损伤情况
A、B和C分别为对照组7、14和21 d的正常气管组织结构。D、E和F分别为MS分离株HI感染后7、14和21 d的气管组织形态。
Figure 4. Histopathology lesions of trachea of 7-day-old SPF chickens infected with HI strain for 7, 14, and 21 days
A–C: Normal tracheal structure of negative control chicken at 7, 14, and 21 days after infection; D–F: Morphological change of the trachea at 7, 14, and 21 days after infection of HI strain, respectively.
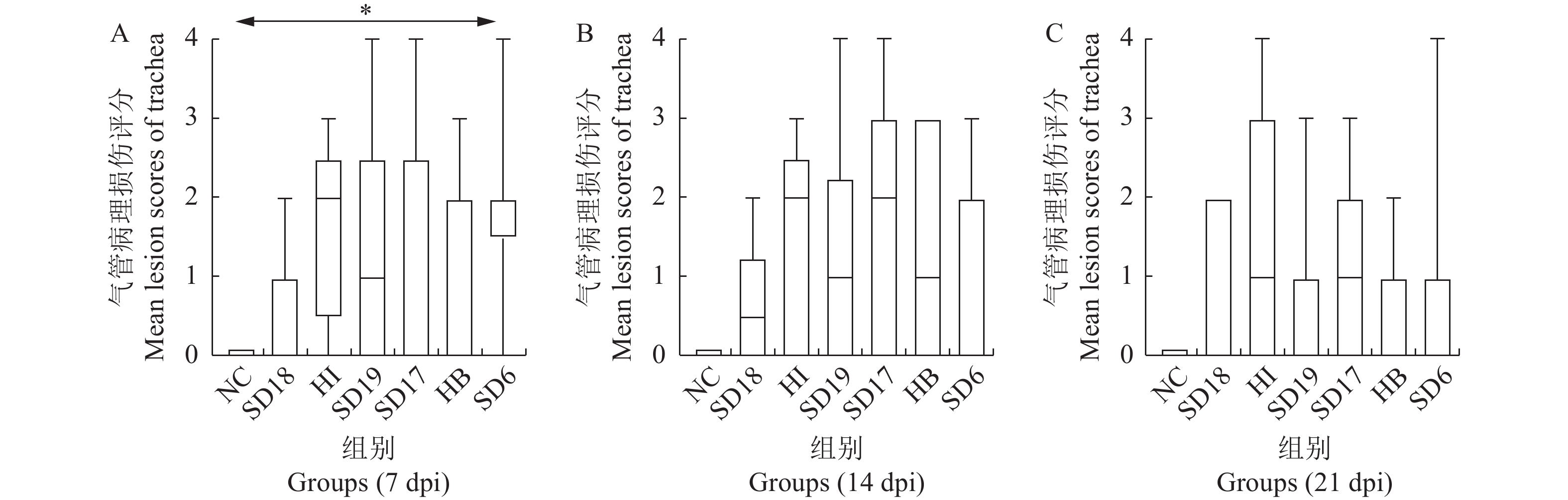
图 5 不同MS分离株感染SPF鸡7 (A)、14 (B)和21 (C) d后气管组织病理学损伤评分(箱线图)
NC为未感染的对照组,数据以非参数的Kruskal-Wallis统计方法进行检验,并进行多重比较,当P< 0.05时表示差异显著,以*表示。
Figure 5. Mean lesion scores of trachea of 7-day-old chickens infected with MS isolates for 7 (A), 14 (B) and 21 (C) days (Box-plot)
NC: No challenge group(control). Data were analyzed using Kruskal-Wallis (for non-parametric data), followed by Dunn’s multiple comparison tests, * P< 0.05 indicates a significant difference.
表 1 不同MS菌株对7日龄SPF鸡感染7、14、21 d后气管病原再分离结果
Table 1. Re-isolation of MS from trachea of 7-old day SPF chickens infected with different MS strains after 7, 14, and 21 days
组别
Group菌株
Strain再分离率
Results of re-isolation感染后7 d
Re-isolation of
7 dpi感染后14 d
Re-isolation of
14 dpi感染后21 d
Re-isolation of
21 dpiG1 未接种No challenge 0/6 0/7 0/7 G2 MS-SD18 0/6(0/3)* 1/7(0/3) 0/7(0/4) G3 MS-HI 3/6(1/3) 5/7(0/3) 5/7(1/4) G4 MS-SD19 1/6(0/3) 6/7(2/3) 2/7(2/4) G5 MS-SD17 0/6(0/3) 3/7(0/3) 5/7(1/4) G6 MS-HB 1/6(1/3) 0/7(0/3) 0/7(0/4) G7 MS-SD6 4/6(0/3) 2/7(3/3) 5/7(1/4) *表中数据为阳性数/检测数(同居鸡的阳性数/检测数)。
Data in the table was number of positive samples/number of tested samples, and number of positive samples/ number of tested samples for cohabitation chickens in parentheses. -
[1] SUN S K, LIN X, CHEN F, et al. Epidemiological investigation of Mycoplasma Synoviae in native chicken breeds in China [J]. BMC Veterinary Research, 2017, 13(1): 115. doi: 10.1186/s12917-017-1029-0 [2] 羊扬, 纪凯丽, 张欣, 等. 广西部分黄羽肉鸡场鸡滑液囊支原体感染的血清学与分子病原学检测 [J]. 中国家禽, 2019, 41(6):73−76. doi: 10.16372/j.issn.1004-6364.2019.06.017YANG Y, JI K L, ZHANG X, et al. Serology and molecular etiology of Mycoplasma synoviae infection in yellow-feather chicken farm in Guangxi [J]. China Poultry, 2019, 41(6): 73−76.(in Chinese) doi: 10.16372/j.issn.1004-6364.2019.06.017 [3] SUI C G, CUI H, JI J, et al. Epidemiological investigations and locally determined genotype diversity of Mycoplasma synoviae in central China from 2017 to 2019 [J]. Poultry Science, 2022, 101(1): 101522. doi: 10.1016/j.psj.2021.101522 [4] VARDAMAN T H, REECE F N, DEATON J W. Effect of Mycoplasma synoviae on broiler performance [J]. Poultry Science, 1973, 52(5): 1909−1912. doi: 10.3382/ps.0521909 [5] KURSA O, PAKUŁA A, TOMCZYK G, et al. Eggshell apex abnormalities caused by two different Mycoplasma synoviae genotypes and evaluation of eggshell anomalies by full-field optical coherence tomography [J]. BMC Veterinary Research, 2019, 15(1): 1. doi: 10.1186/s12917-018-1758-8 [6] LOCKABY S B, HOERR F J, LAUERMAN L H, et al. Pathogenicity of Mycoplasma synoviae in broiler chickens [J]. Veterinary Pathology, 1998, 35(3): 178−190. doi: 10.1177/030098589803500303 [7] KLEVEN S H, FERGUSON-NOEL N. Mycoplasma synoviae infection [A]// SAIF Y M, FADLY A H, GLISSON J R, MCDOUGALD J R, NOLAN N K, SWAYNE D E. Diseases of poultry[M]. 12th Edition. Ames, Iowa, USA: Blackwell Publishing, 2008: 845-856. [8] 侯博, 王晨燕, 邵国青. 滑液囊支原体不同感染途径的致病力比较 [J]. 微生物学通报, 2021, 48(8):2704−2713. doi: 10.13344/j.microbiol.china.210255HOU B, WANG C Y, SHAO G Q. Comparison of pathogenicity to Mycoplasma synoviae with different infection routs [J]. Microbiology China, 2021, 48(8): 2704−2713.(in Chinese) doi: 10.13344/j.microbiol.china.210255 [9] RAVIV Z, FERGUSON-NOEL N, LAIBINIS V, et al. Role of Mycoplasma synoviae in commercial layer Escherichia coli peritonitis syndrome [J]. Avian Diseases, 2007, 51(3): 685−690. doi: 10.1637/0005-2086(2007)51[685:ROMSIC]2.0.CO;2 [10] LANDMAN W J M, FEBERWEE A. Aerosol-induced Mycoplasma synoviae arthritis: The synergistic effect of infectious bronchitis virus infection [J]. Avian Pathology:Journal of the WVPA, 2004, 33(6): 591−598. doi: 10.1080/03079450400013170 [11] FEBERWEE A, MORROW C J, GHORASHI S A, et al. Effect of a live Mycoplasma synoviae vaccine on the production of eggshell apex abnormalities induced by a M. synoviae infection preceded by an infection with infectious bronchitis virus D1466 [J]. Avian Pathology:Journal of the WVPA, 2009, 38(5): 333−340. doi: 10.1080/03079450903183652 [12] AHMAD ROUSSAN D, KHAWALDEH G, ALI SHAHEEN I. A survey of Mycoplasma gallisepticum and Mycoplasma synovaie with avian influenza H9 subtype in meat-type chicken in Jordan between 2011–2015 [J]. Poultry Science, 2015, 94(7): 1499−1503. doi: 10.3382/ps/pev119 [13] 中华人民共和国农业部. 禽支原体PCR检测方法: NY/T 553—2015[S]. 北京: 中国农业出版社, 2015. [14] EL-GAZZAR M M, WETZEL A N, RAVIV Z. The genotyping potential of the Mycoplasma synoviae vlhA gene [J]. Avian Diseases, 2012, 56(4): 711−719. doi: 10.1637/10200-041212-Reg.1 [15] WANG C Y, LUO Z B, SHAO G Q, et al. Genetic and pathogenic characteristics of a novel infectious bronchitis virus strain in genogroup VI (CK/CH/FJ/202005) [J]. Veterinary Microbiology, 2022, 266: 109352. doi: 10.1016/j.vetmic.2022.109352 [16] XUE J, XU M Y, MA Z J, et al. Serological investigation of Mycoplasma synoviae infection in China from 2010 to 2015 [J]. Poultry Science, 2017, 96(9): 3109−3112. doi: 10.3382/ps/pex134 [17] EWING M L, COOKSON K C, PHILLIPS R A, et al. Experimental infection and transmissibility of Mycoplasma synoviae with delayed serologic response in chickens [J]. Avian Diseases, 1998, 42(2): 230−238. doi: 10.2307/1592472 [18] KHIARI A B, MARDASSI B B. Characterization of the antigenic and functional domains of a Mycoplasma synoviae variant vlhA gene [J]. Veterinary Microbiology, 2012, 156(3/4): 322−329. [19] KLEVEN S H, KING D D, ANDERSON D P. Airsacculitis in broilers from Mycoplasma synoviae: Effect on air-sac lesions of vaccinating with infectious bronchitis and Newcastle virus [J]. Avian Diseases, 1972, 16(4): 915−924. doi: 10.2307/1588772 [20] 宫晓炜, 陈启伟, Ferguson-Noel Naola, 等. 3株禽滑液囊支原体分离株致病性的比较和评价 [J]. 微生物学通报, 2019, 46(4):790−797. doi: 10.13344/j.microbiol.china.180339GONG X W, CHEN Q W, FERGUSONNOEL N L, et al. Comparison the pathogenicity of three Mycoplasma synoviae isolates from broiler flocks [J]. Microbiology China, 2019, 46(4): 790−797.(in Chinese) doi: 10.13344/j.microbiol.china.180339 -






 下载:
下载: