Cloning and Expression of MeERF1.2 in Cassava
-
摘要:
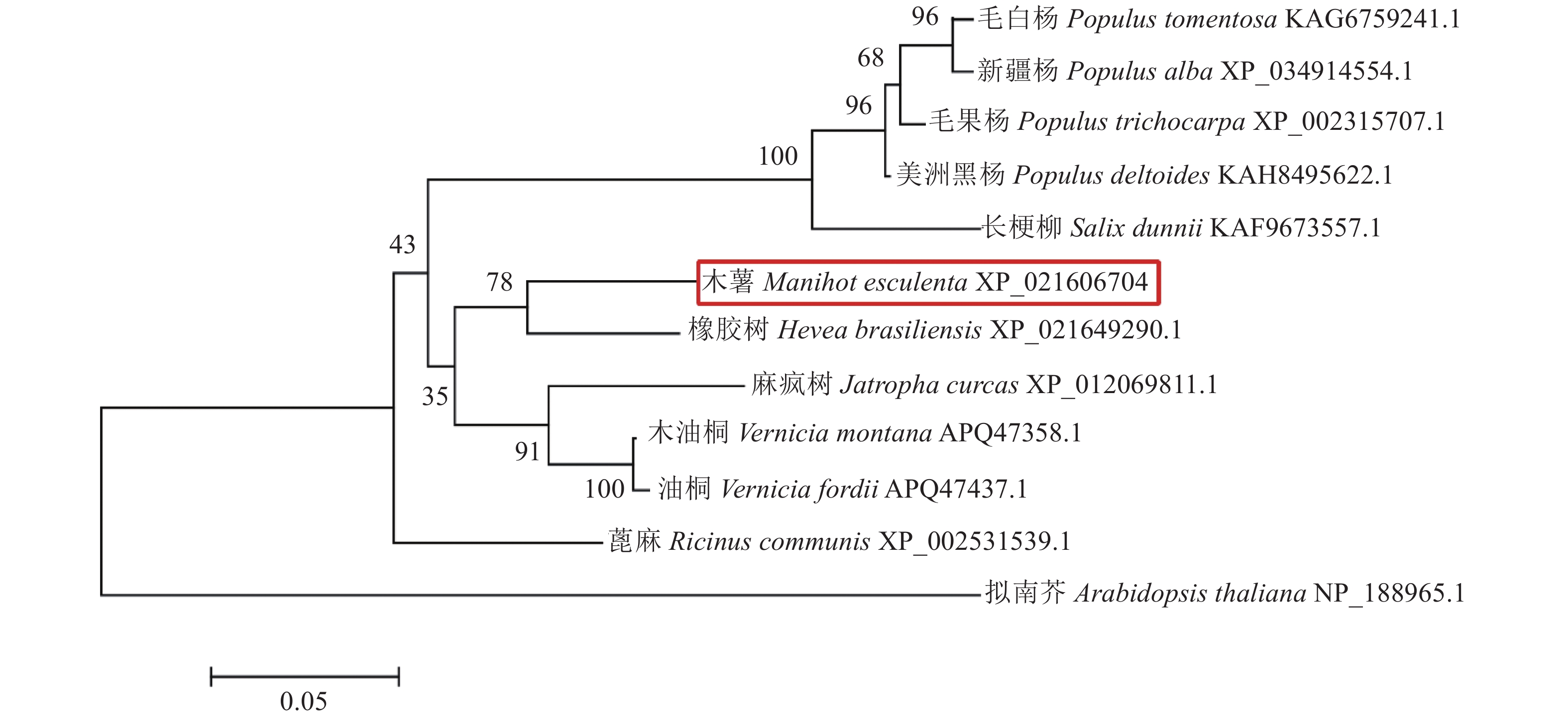
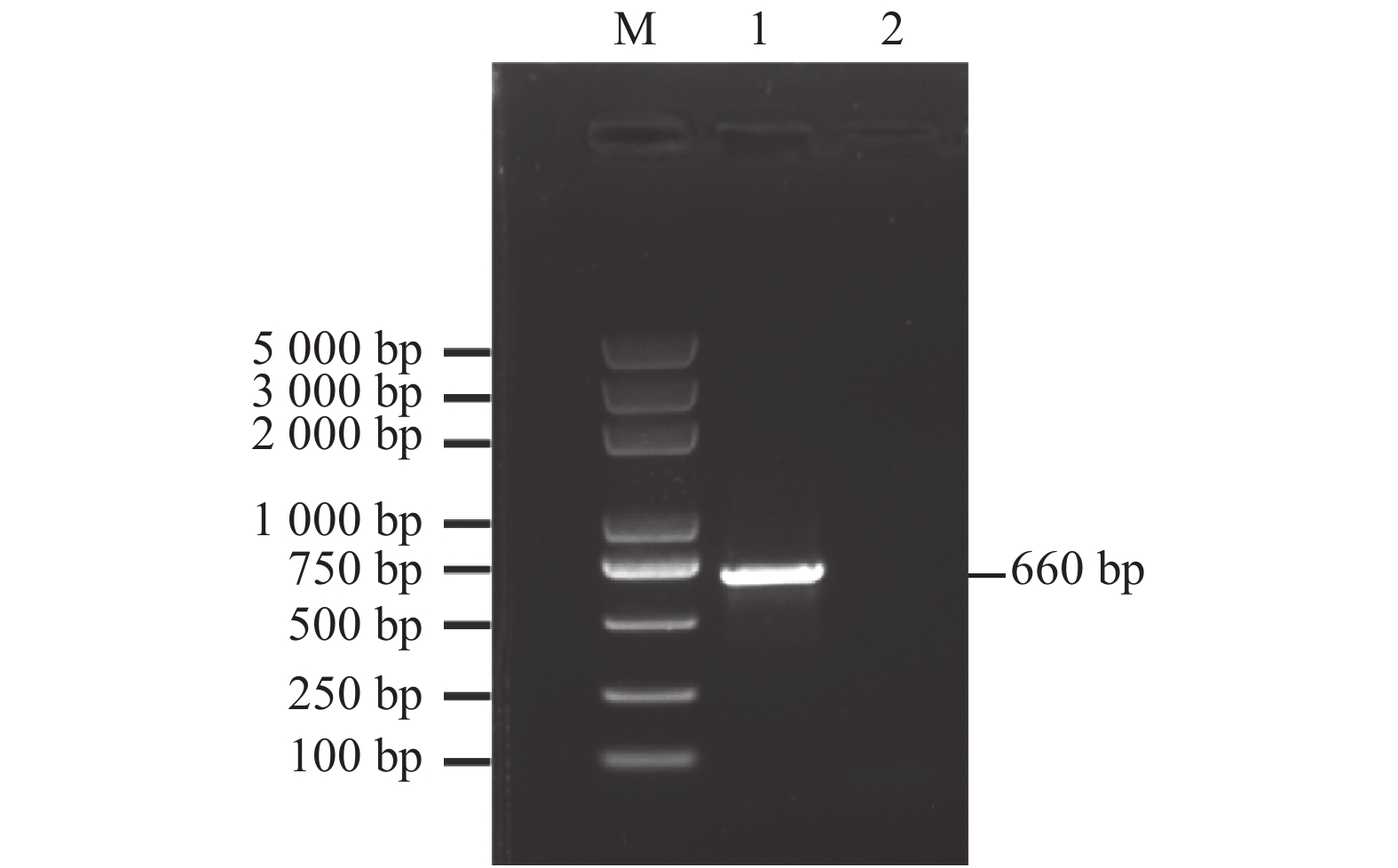
目的 乙烯响应因子(Ethylene response factor,ERF)是乙烯信号转导通路的重要成员,克隆并分析其在木薯块根采后生理性变质(Post-harvest physiological deterioration,PPD)过程中的表达情况,能为进一步研究乙烯信号在木薯PPD过程中的功能提供参考。 方法 以木薯栽培品种华南8号(SC8)为材料,采用RT-PCR技术克隆木薯MeERF1.2基因,对其进行相关生物信息学分析,如遗传进化关系、结构域、蛋白质结构预测、理化性质等。对MeERF1.2基因在细胞中的亚细胞定位进行确认,并用qRT-PCR技术分析MeERF1.2基因在木薯块根PPD过程中的表达水平。 结果 克隆得到的MeERF1.2基因全长为660 bp,编码的氨基酸残基数为219,分子量和等电点分别为25.04 kD和5.61,含有AP2家族结构域,和橡胶HbERF1B-like的亲缘关系最近,序列相似性达到88.74%。MeERF1.2基因定位于细胞核。和对照0 h相比,MeERF1.2基因的表达量在木薯块根的采后过程中表现为显著上升趋势,即MeERF1.2基因的表达受到PPD过程的诱导。 结论 克隆得到的MeERF1.2基因包含ERF基因家族的保守结构域,属于ERF基因家族;MeERF1.2基因的表达在采后过程中受到诱导,可能参与了木薯块根的PPD过程,为后续进一步分析乙烯信号转导通路在PPD过程中的作用奠定了基础。 Abstract:Objective Critical factors in the ethylene signal transduction pathway involving the post-harvest physiological deterioration (PPD) of cassava were investigated. Method One of the possible ethylene response factors (ERFs) in Manihot esculenta cv.SC8, MeERF1.2, was cloned using RT-PCR to analyze the expressions during PPD. Physicochemical properties, conserved domain, genetic evolutionary relationship, and protein structure of the gene were studied with bioinformatics tools. Distribution of MeERF1.2 in the plant cells was verified by online software and subcellular localization. Result MeERF1.2 had an ORF of 660 bp encoded 188 amino acids with a molecular weight of 25.04 kD and a pI of 5.61. The protein contained an AP2 domain showing a high sequence similarity of 88.74% with the HbERF1B-like gene. It was localized in the nucleus and significantly upregulated during PPD from 0 to 6, 12, and 48 h displaying an apparent induction trend in the process. Conclusion MeERF1.2 was one of the ERFs induced in cassava tubers during PPD and played a vital role in the signal transduction pathway of the plant. -
Key words:
- Cassava /
- ethylene /
- ERF /
- ROS /
- post-harvest physiological deterioration
-
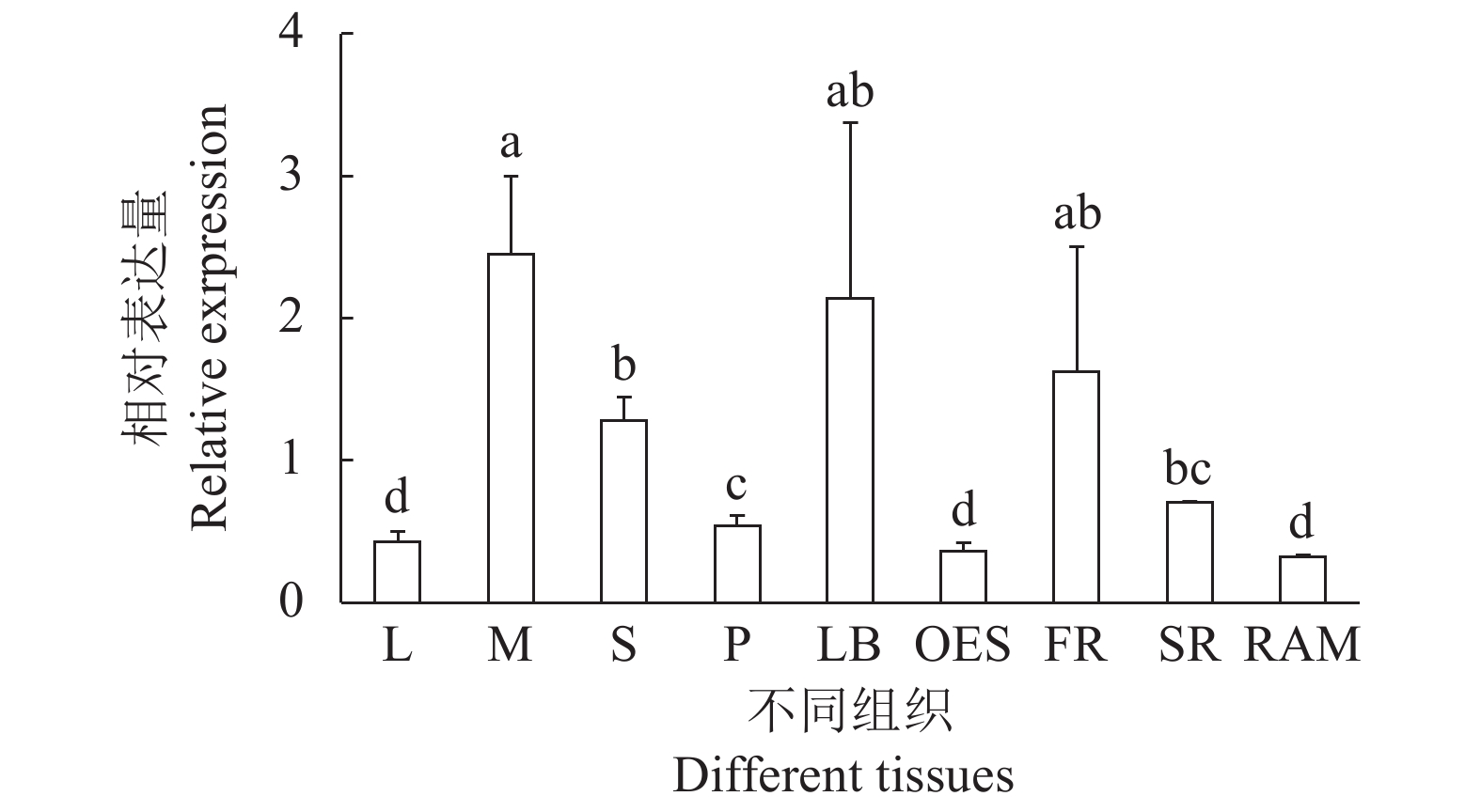
图 6 MeERF1.2在不同组织中的表达量
L:叶,M:中脉,S:茎,P:叶柄,LB:侧芽,OES:分化胚组织,FR:须根,SR:根,RAM:根顶端分生组织。不同小写字母表明在Duncan’s多重比较中差异显著(P<0.05)
Figure 6. Expressions of MeERF1.2 in tissues
L: leaf; M: midvein; S: stem; P: petiole; LB: lateral bud; OES: organized embryogenic structure; FR: fibrous root; SR: storage root; RAM: root apical meristem. Data with different letters indicate significant differences based on Duncan's multiple range tests (P<0.05).
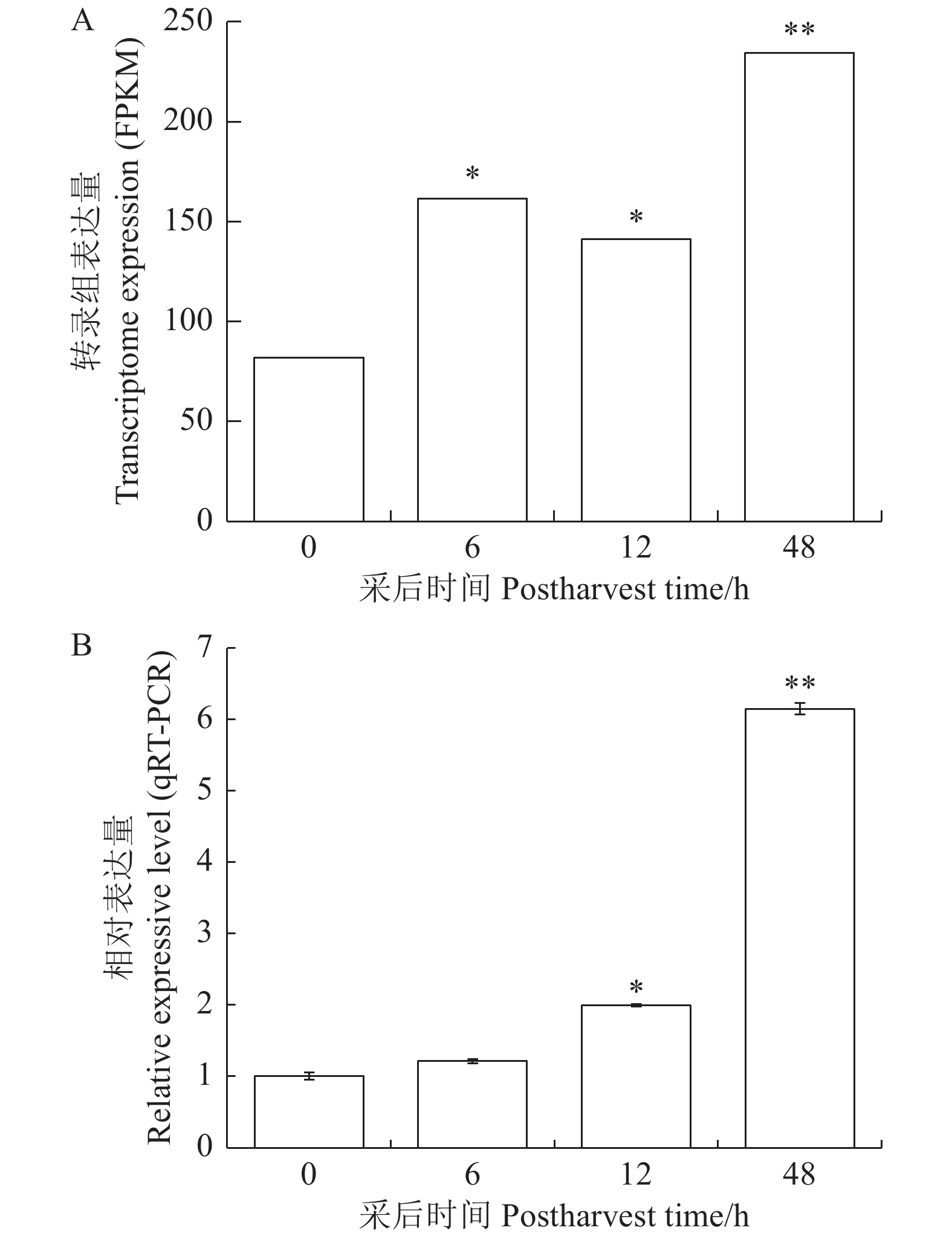
图 7 MeERF1.2基因在PPD中的表达变化
A MeERF1.2基因在采后腐烂过程中转录组测序表达量;B: MeERF1.2基因在采后腐烂过程中实时定量PCR检测结果;*和**分别代表与0 h差异显著(P<0.05)和极显著(P<0.01)。
Figure 7. Expressions of MeERF1.2 during PPD
A: expression of MeERF1.2 during cassava PPD shown by transcriptome sequencing; B: expression of MeERF1.2 during cassava PPD shown by qRT-PCR; * and ** represent significantly different from control (0 h) at P<0.05 and P<0.01, respectively.
表 1 扩增引物序列
Table 1. Primers and their sequences
引物名称
Primer引物序列
Primer sequence引物用途
Primer usageMeERF1.2-F 5′-ATGGATTCCTCCATCTTTCATTCT-3′ 基因克隆 MeERF1.2-R 5′-CCAAGGTCTTATAGCATTCTCAGAT-3′ BiMeERF1.2-F 5′-AGTGGTCTCTGTCCAGTCCTATGGATTCCTCCATCTTT-3′ 亚细胞定位 BiMeERF1.2-R 5′-GGTCTCAGCAGACCACAAGTCCAAGGTCTTATAGCATT-3′ qMeERF1.2-F 5′-GAGCTGGGGCTGTACTCAAT-3′ 荧光定量PCR qMeERF1.2-R 5′-CAGGTGAGCACCCTTCTTCT-3′ MeEF1-F 5′-TGAACCACCCTGGTCAGATTGGAA-3′ 内参基因 MeEF1-R 5′-AACTTGGGCTCCTTCTCAAGCTCT-3′ -
[1] 张鹏, 杨俊, 周文智, 等. 能源木薯高淀粉抗逆分子育种研究进展与展望 [J]. 生命科学, 2014, 26(5):465−473.ZHANG P, YANG J, ZHOU W Z, et al. Progress and perspective of cassava molecular breeding for bioenergy development [J]. Chinese Bulletin of Life Sciences, 2014, 26(5): 465−473.(in Chinese) [2] HU W, KONG H, GUO Y L, et al. Comparative physiological and transcriptomic analyses reveal the actions of melatonin in the delay of postharvest physiological deterioration of cassava [J]. Frontiers in Plant Science, 2016, 7: 736. [3] 郭靖, 章玉香, 黄芷颐, 等. 木薯MePYL12基因克隆及采后生理性变质过程的表达分析 [J]. 福建农业学报, 2021, 36(1):17−23.GUO J, ZHANG Y X, HUANG Z Y, et al. Cloning and post-harvest physiological deterioration expression of MePYL12 of cassava [J]. Fujian Journal of Agricultural Sciences, 2021, 36(1): 17−23.(in Chinese) [4] WESTBY A. Cassava utilization, storage and small-scale processing[M]//Cassava: biology, production and utilization. UK: CABI Publishing, 2001: 281-300. [5] 马秋香, 许佳, 乔爱民, 等. 木薯储藏根采后生理性变质研究进展 [J]. 热带亚热带植物学报, 2009, 17(3):309−314.MA Q X, XU J, QIAO A M, et al. Current progress in studies on post-harvest physiological deterioration of cassava storage roots [J]. Journal of Tropical and Subtropical Botany, 2009, 17(3): 309−314.(in Chinese) [6] IYER S, MATTINSON D S, FELLMAN J K. Study of the early events leading to cassava root postharvest deterioration [J]. Tropical Plant Biology, 2010, 3(3): 151−165. doi: 10.1007/s12042-010-9052-3 [7] HERSHKOVITZ V, FRIEDMAN H, GOLDSCHMIDT E E, et al. Induction of ethylene in avocado fruit in response to chilling stress on tree [J]. Journal of Plant Physiology, 2009, 166(17): 1855−1862. doi: 10.1016/j.jplph.2009.05.012 [8] 赵赫, 陈受宜, 张劲松. 乙烯信号转导与植物非生物胁迫反应调控研究进展 [J]. 生物技术通报, 2016, 32(10):1−10.ZHAO H, CHEN S Y, ZHANG J S. Ethylene signaling pathway in regulating plant response to abiotic stress [J]. Biotechnology Bulletin, 2016, 32(10): 1−10.(in Chinese) [9] 施怡婷, 杨淑华. 中国科学家在乙烯信号转导领域取得突破性进展 [J]. 植物学报, 2016, 51(3):287−289.SHI Y T, YANG S H. Chinese scientists made breakthrough in study on ethylene signaling transduction in plants [J]. Chinese Bulletin of Botany, 2016, 51(3): 287−289.(in Chinese) [10] REN M Y, FENG R J, SHI H R, et al. Expression patterns of members of the ethylene signaling-related gene families in response to dehydration stresses in cassava [J]. PLoS One, 2017, 12(5): e0177621. doi: 10.1371/journal.pone.0177621 [11] CAsO W H, LIU J, CHEN T, et al. Ethylene receptor signaling and plant salt-stress responses[C]//RAMINA A, CHANG C, GIOVANNONI J, et al. Advances in Plant Ethylene Research. Dordrecht: Springer, 2007: 333-339. [12] 于延文, 黄荣峰. 乙烯与植物抗逆性 [J]. 中国农业科技导报, 2013, 15(2):70−75.YU Y W, HUANG R F. Ethylene and plant resistance to adversity [J]. Journal of Agricultural Science and Technology, 2013, 15(2): 70−75.(in Chinese) [13] DESIKAN R, LAST K, HARRETT-WILLIAMS R, et al. Ethylene-induced stomatal closure in Arabidopsis occurs via AtrbohF-mediated hydrogen peroxide synthesis [J]. The Plant Journal:for Cell and Molecular Biology, 2006, 47(6): 907−916. doi: 10.1111/j.1365-313X.2006.02842.x [14] 杨洋. 拟南芥乙烯合成因子依赖ROS代谢调节盐胁迫反应[D]. 开封: 河南大学, 2011.YANG Y. The ethylene biosynthesis factors mediate Arabidopsis tolerance to NaCl stress in dependence on ROS metabolism[D]. Kaifeng: Henan University, 2011. (in Chinese) [15] MA Y R, YANG M N, WANG J J, et al. Application of exogenous ethylene inhibits postharvest peel browning of ‘Huangguan’ pear [J]. Frontiers in Plant Science, 2017, 7: 2029. [16] BLEECKER A B, KENDE H. Ethylene: A gaseous signal molecule in plants [J]. Annual Review of Cell and Developmental Biology, 2000, 16: 1−18. doi: 10.1146/annurev.cellbio.16.1.1 [17] ALONSO J M, STEPANOVA A N, SOLANO R, et al. Five components of the ethylene-response pathway identified in a screen for weak ethylene-insensitive mutants in Arabidopsis [J]. Proceedings of the National Academy of Sciences of the United States of America, 2003, 100(5): 2992−2997. doi: 10.1073/pnas.0438070100 [18] ZHAO Q, GUO H W. Paradigms and paradox in the ethylene signaling pathway and interaction network [J]. Molecular Plant, 2011, 4(4): 626−634. doi: 10.1093/mp/ssr042 [19] 史庆玲, 李忠峰, 董永彬, 等. 植物乙烯信号转导通路及其相关基因的研究进展 [J]. 生物技术进展, 2019, 9(5):449−454.SHI Q L, LI Z F, DONG Y B, et al. Progress on ethylene signal transduction pathway and related genes in plants [J]. Current Biotechnology, 2019, 9(5): 449−454.(in Chinese) [20] 葛宝宇, 林轶, 侯和胜. ERF类转录因子的结构与功能 [J]. 安徽农学通报, 2007, 13(20):32−35. doi: 10.3969/j.issn.1007-7731.2007.20.013GE B Y, LIN Y, HOU H S. Structure and function of ERF transcription factors [J]. Anhui Agricultural Science Bulletin, 2007, 13(20): 32−35.(in Chinese) doi: 10.3969/j.issn.1007-7731.2007.20.013 [21] NAKANO T, SUZUKI K, FUJIMURA T, et al. Genome-wide analysis of the ERF gene family in Arabidopsis and rice [J]. Plant Physiology, 2006, 140(2): 411−432. doi: 10.1104/pp.105.073783 [22] SHOJI T, MISHIMA M, HASHIMOTO T. Divergent dna-binding specificities of a group of ethylene response factor transcription factors involved in plant defense [J]. Plant Physiology, 2013, 162(2): 977−990. doi: 10.1104/pp.113.217455 [23] ABIRI R, SHAHARUDDIN N A, MAZIAH M, et al. Role of ethylene and the APETALA 2/ethylene response factor superfamily in rice under various abiotic and biotic stress conditions [J]. Environmental and Experimental Botany, 2017, 134: 33−44. doi: 10.1016/j.envexpbot.2016.10.015 [24] SOLANO R, STEPANOVA A, CHAO Q, et al. Nuclear events in ethylene signaling: A transcriptional cascade mediated by ETHYLENE-INSENSITIVE3 and ETHYLENE-RESPONSE-FACTOR1 [J]. Genes & Development, 1998, 12(23): 3703−3714. [25] QIN F, SAKUMA Y, LI J, et al. Cloning and functional analysis of a novel DREB1/CBF transcription factor involved in cold-responsive gene expression in Zea mays L [J]. Plant and Cell Physiology, 2004, 45(8): 1042−1052. doi: 10.1093/pcp/pch118 [26] VANDERSCHUREN H, NYABOGA E, POON J S, et al. Large-scale proteomics of the cassava storage root and identification of a target gene to reduce postharvest deterioration [J]. The Plant Cell, 2014, 26(5): 1913−1924. doi: 10.1105/tpc.114.123927 [27] 张云川, 曹天澳, 雷骥良, 等. 橡胶草ERF3基因的克隆、亚细胞定位及表达分析 [J]. 植物生理学报, 2021, 57(6):1261−1270.ZHANG Y C, CAO T A, LEI J L, et al. Cloning, subcellular localization and expression analysis of TkERF3 in Taraxacum kok-saghyz [J]. Plant Physiology Journal, 2021, 57(6): 1261−1270.(in Chinese) [28] JUNG H, CHUNG P J, PARK S H, et al. Overexpression of OsERF48 causes regulation of OsCML16, a calmodulin-like protein gene that enhances root growth and drought tolerance [J]. Plant Biotechnology Journal, 2017, 15(10): 1295−1308. doi: 10.1111/pbi.12716 [29] LEE D K, YOON S, KIM Y S, et al. Rice OsERF71-mediated root modification affects shoot drought tolerance [J]. Plant Signaling & Behavior, 2017, 12(1): e1268311. [30] ZHANG G Y, CHEN M, LI L C, et al. Overexpression of the soybean GmERF3 gene, an AP2/ERF type transcription factor for increased tolerances to salt, drought, and diseases in transgenic tobacco [J]. Journal of Experimental Botany, 2009, 60(13): 3781−3796. doi: 10.1093/jxb/erp214 [31] CHARFEDDINE M, CHARFEDDINE S, GHAZALA I, et al. Investigation of the response to salinity of transgenic potato plants overexpressing the transcription factor StERF94 [J]. Journal of Biosciences, 2019, 44(6): 141. doi: 10.1007/s12038-019-9959-2 [32] SHIN S Y, PARK M H, CHOI J W, et al. Gene network underlying the response of harvested pepper to chilling stress [J]. Journal of Plant Physiology, 2017, 219: 112−122. doi: 10.1016/j.jplph.2017.10.002 -






 下载:
下载: